Structure and Bonding
Matthew Williams
||11 min readAllotropyCovalent BondingCrystal StructuresCSEC ChemistryIonic BondingMetallic BondingPaper 01Paper 02Section A
Ionic, covalent, and metallic bonding, dot-and-cross diagrams, predicting bond type from atomic structure, types of crystal structures, properties of ionic and molecular solids, structure and uses of sodium chloride, diamond and graphite, and allotropy.
The melting point, solubility, electrical conductivity, and hardness of a substance all follow directly from how its particles are bonded and arranged. This page covers how bonds form, what structures result, and the properties those structures produce.
Why Atoms Bond
Most atoms are chemically reactive because their outer electron shells are incomplete. Atoms achieve stability — an arrangement matching the electron configuration of a noble gas — by:
- losing electrons (forming positive ions)
- gaining electrons (forming negative ions)
- sharing electrons (forming covalent bonds)
Noble gases (Group 0) are stable and unreactive precisely because their outer shells are already full.
Ionic Bonding
Ionic bonding is the strong electrostatic attraction between oppositely charged ions. It forms when one or more electrons are transferred from a metal atom to a non-metal atom. The metal becomes a positive cation; the non-metal becomes a negative anion. Both achieve a full outer shell.
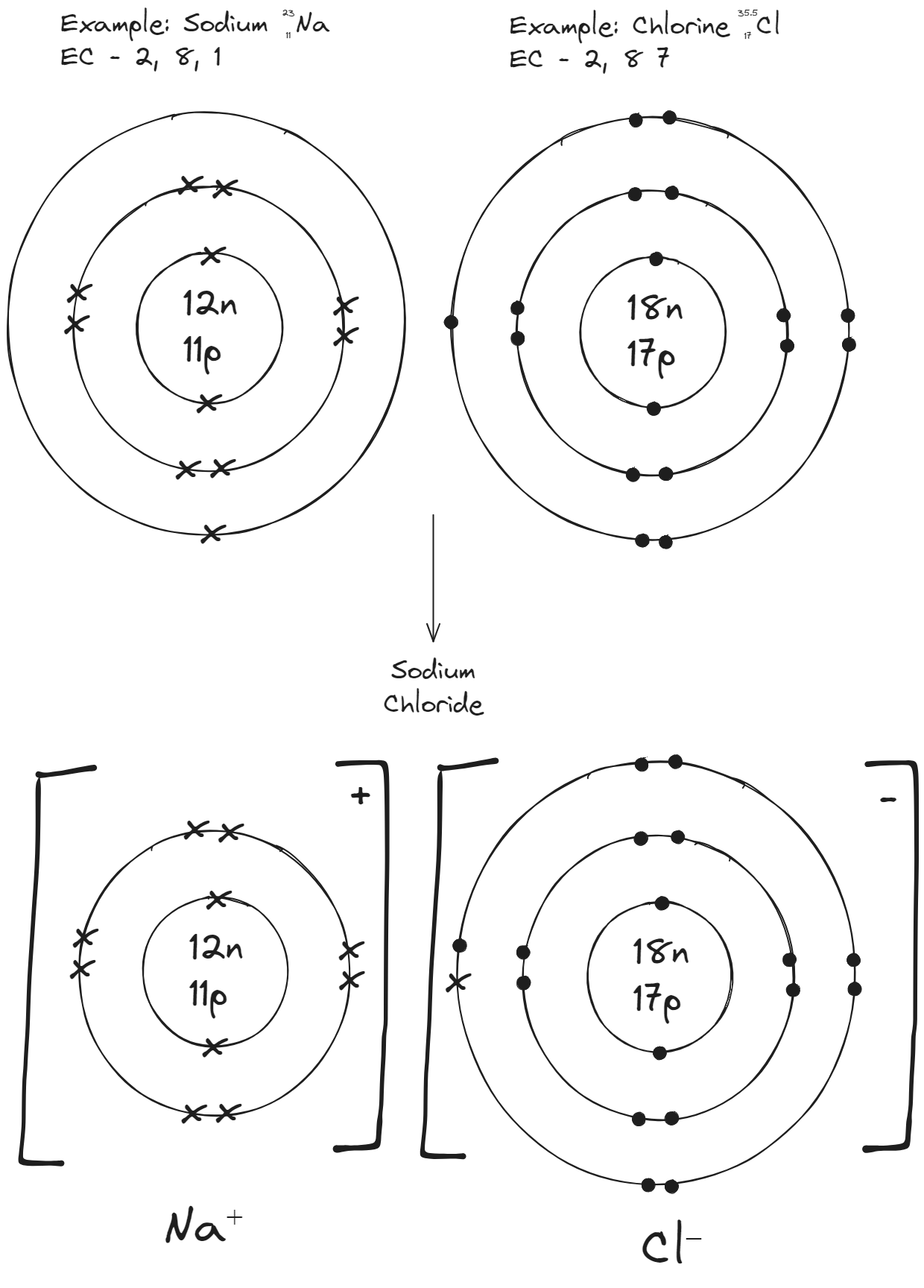
Formation of Sodium Chloride
Sodium (2,8,1) has one electron in its outer shell. Chlorine (2,8,7) needs one more to complete its outer shell.
- Na → Na⁺ + e⁻
- Cl + e⁻ → Cl⁻
- The oppositely charged ions attract each other — this electrostatic attraction is the ionic bond.
Other common ionic compounds:
| Compound | Formula | Ions |
|---|---|---|
| Magnesium oxide | MgO | Mg²⁺ and O²⁻ |
| Calcium chloride | CaCl₂ | Ca²⁺ and 2Cl⁻ |
| Aluminium oxide | Al₂O₃ | 2Al³⁺ and 3O²⁻ |
| Potassium bromide | KBr | K⁺ and Br⁻ |
Predicting Bond Type
The type of bond that forms depends on the types of atoms involved:
| Combination | Bond type | Reason | Example |
|---|---|---|---|
| Metal + non-metal | Ionic | Metal loses electrons; non-metal gains them | NaCl, MgO, CaBr₂ |
| Non-metal + non-metal | Covalent | Neither atom easily loses electrons; they share instead | H₂O, CO₂, NH₃ |
Formulae of Ions and Molecules
An ionic compound formula is written so that the total positive charge equals the total negative charge — the compound is electrically neutral overall. To balance charges, use the charge of each ion as the subscript of the other (cross-multiply, then simplify).
Common ions to know:
| Cation | Formula | Anion | Formula |
|---|---|---|---|
| Sodium | Na⁺ | Chloride | Cl⁻ |
| Potassium | K⁺ | Bromide | Br⁻ |
| Magnesium | Mg²⁺ | Oxide | O²⁻ |
| Calcium | Ca²⁺ | Sulfide | S²⁻ |
| Aluminium | Al³⁺ | Nitride | N³⁻ |
| Ammonium | NH₄⁺ | Hydroxide | OH⁻ |
| Nitrate | NO₃⁻ | ||
| Carbonate | CO₃²⁻ | ||
| Sulfate | SO₄²⁻ |
Example
Aluminium oxide: Al is 3+ and O is 2−. To balance: 2 × Al³⁺ gives 6+; 3 × O²⁻ gives 6−. Formula: Al₂O₃.
Covalent Bonding
Covalent bonding forms when two non-metal atoms share one or more pairs of electrons. Each shared pair is one covalent bond. Both atoms achieve a full outer shell through sharing. Electrons not involved in bonding sit as lone pairs on each atom.
| Bond type | Shared pairs | Examples |
|---|---|---|
| Single bond | 1 | H₂, HCl, H₂O, NH₃, CH₄ |
| Double bond | 2 | O₂, CO₂ |
| Triple bond | 3 | N₂ |
Dot-and-Cross Diagrams
In a dot-and-cross diagram, one atom's electrons are shown as dots and the other's as crosses. Shared pairs sit between the two atoms; lone pairs sit on the outer edge of each atom.

[DIAGRAM PLACEHOLDER: Dot-and-cross diagrams for H₂, Cl₂, HCl, H₂O, NH₃, CH₄, O₂ (double bond), N₂ (triple bond), and CO₂ (two double bonds)]
Key molecules to know for CSEC:
| Molecule | Bond type | Lone pairs |
|---|---|---|
| H₂ | 1 single bond | None |
| Cl₂ | 1 single bond | 3 lone pairs per Cl |
| H₂O | 2 single bonds | 2 lone pairs on O |
| NH₃ | 3 single bonds | 1 lone pair on N |
| CH₄ | 4 single bonds | None |
| O₂ | 1 double bond | 2 lone pairs per O |
| CO₂ | 2 double bonds | 2 lone pairs per O |
| N₂ | 1 triple bond | 1 lone pair per N |
Metallic Bonding
In a metal, atoms release their valence electrons into a shared pool. The result is a regular arrangement of positive metal cations surrounded by a sea of delocalised electrons free to move throughout the entire structure.
Metallic bonding is the electrostatic attraction between those positive cations and the mobile electron sea.
[DIAGRAM PLACEHOLDER: Metallic lattice — rows of positive cations surrounded by a sea of delocalised (mobile) electrons]
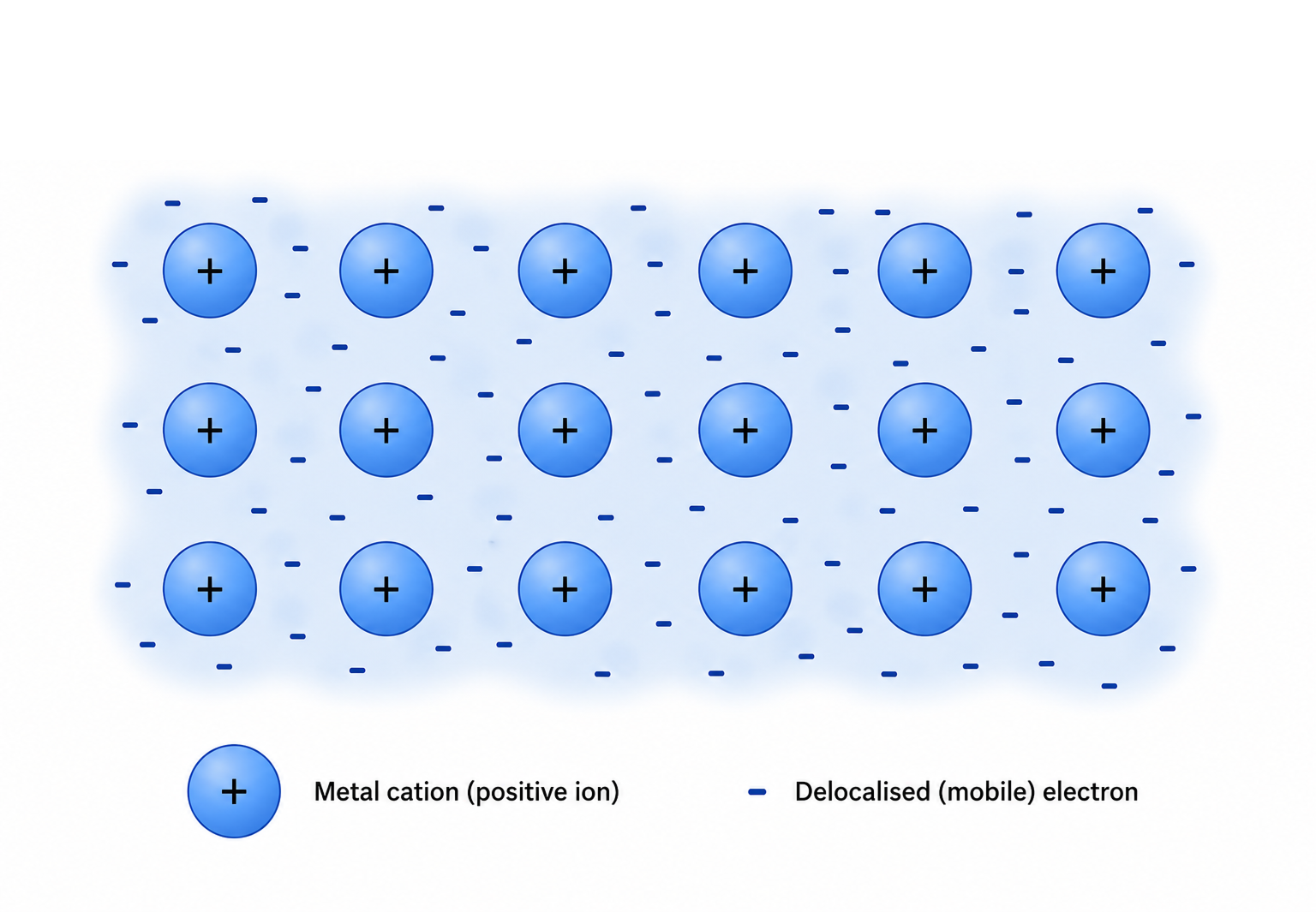
The properties of metals follow directly from this structure:
| Property | Explanation |
|---|---|
| High melting point | Strong attraction between cations and delocalised electrons requires substantial energy to overcome |
| Good electrical conductor | Delocalised electrons carry charge freely through the structure |
| Good thermal conductor | Delocalised electrons transfer kinetic energy rapidly |
| Malleable | Layers of cations slide over each other while the electron sea maintains the bond |
| Ductile | Atoms rearrange without the metallic bonds breaking |
| Shiny lustre | Delocalised electrons reflect light |
Types of Crystal Structures
Solids form three main types of crystal structure, with very different properties.
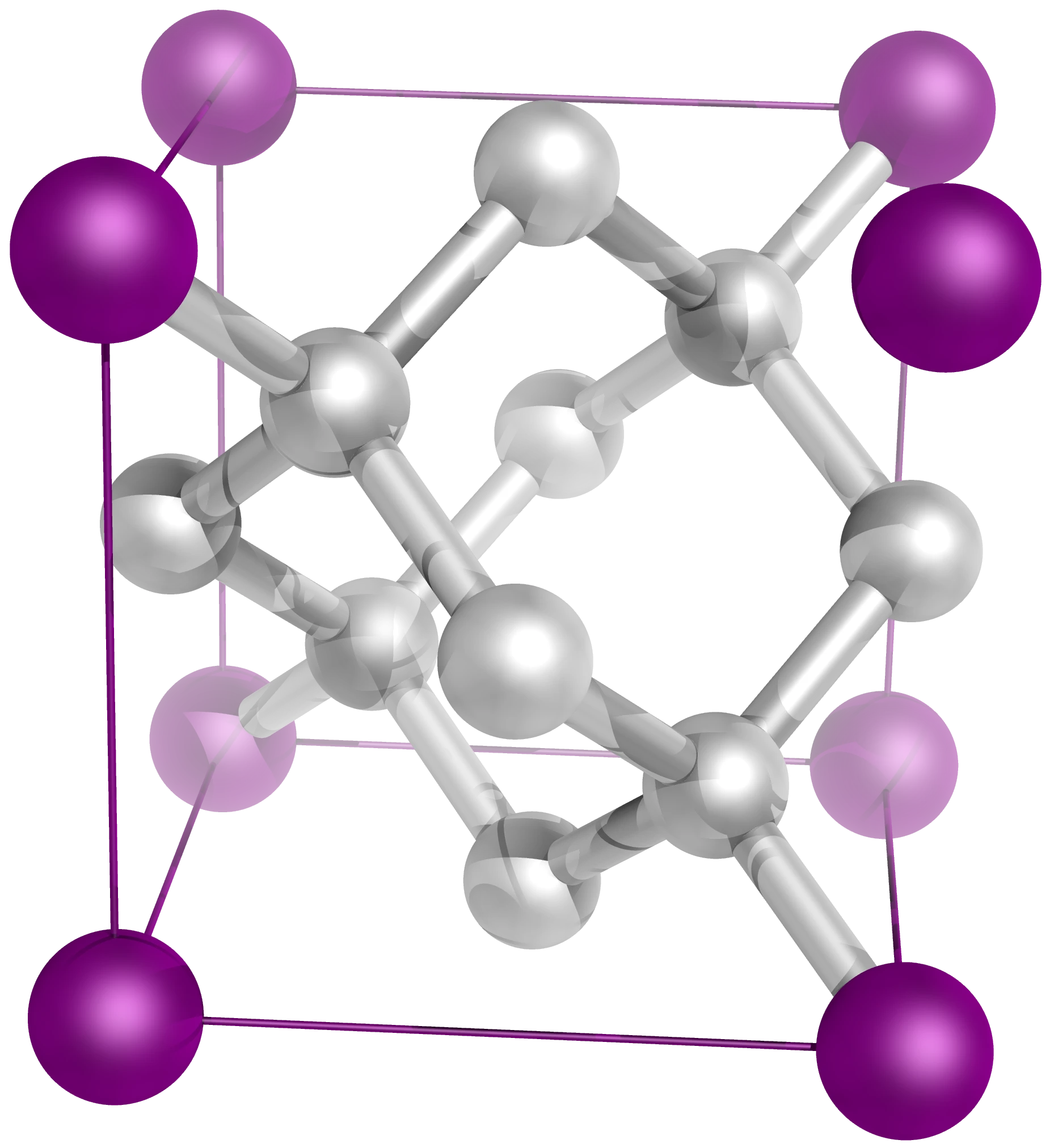
Ionic Crystals
An ionic crystal is a giant three-dimensional lattice of alternating positive and negative ions held together by strong electrostatic attractions in all directions. There are no separate molecules — the entire crystal is one continuous ionic structure.
In sodium chloride, each Na⁺ is surrounded by 6 Cl⁻ ions, and each Cl⁻ is surrounded by 6 Na⁺ ions.

Key: purple spheres = Na⁺ ions, green spheres = Cl⁻ ions
Simple Molecular Crystals
These are composed of small covalent molecules held in the solid state by weak intermolecular forces (van der Waals forces). The covalent bonds within each molecule are strong, but the attractions between molecules are weak. Examples: iodine (I₂), ice (H₂O), dry ice (CO₂), wax.
Giant Molecular (Giant Covalent) Structures
All atoms are joined by covalent bonds throughout the entire solid — there are no separate small molecules. Diamond, graphite, and silicon dioxide all have this structure.
Ionic versus Molecular Solids
| Property | Ionic solids | Simple molecular solids |
|---|---|---|
| Melting point | High — strong ionic bonds throughout the lattice | Low — only weak intermolecular forces to overcome |
| Electrical conductivity | Conduct when molten or dissolved (ions become mobile); solid does not conduct | Do not conduct (no mobile ions or free electrons) |
| Solubility in water | Usually soluble — water molecules surround and separate the ions | Usually insoluble (especially non-polar molecules) |
| Solubility in organic solvents | Usually insoluble | Often soluble |
| Physical character | Hard, brittle crystals | Soft or waxy solids, liquids, or gases |
Exam Tip
A very common exam error is saying ionic compounds conduct electricity in the solid state. They do not — ions are fixed in the lattice. Conductivity only occurs when molten or dissolved, because that is when ions become free to move and carry charge.
Structure, Properties, and Uses
Sodium Chloride (NaCl)
Sodium chloride has a giant ionic lattice. Strong electrostatic attractions between Na⁺ and Cl⁻ ions in all directions give it a high melting point (801 °C). It is hard but brittle — when layers are forced to shift, like charges align, causing sudden repulsion that shatters the crystal. NaCl dissolves readily in water and conducts electricity when molten or dissolved.
Uses: food seasoning and preservation, production of chlorine and sodium hydroxide by electrolysis, de-icing roads.
Diamond
Diamond is a carbon allotrope in which every carbon atom forms four covalent bonds to four other carbon atoms in a giant three-dimensional tetrahedral network. There are no double bonds and no free electrons.
| Property | Reason |
|---|---|
| Extremely hard | Every atom is locked in place by four strong covalent bonds throughout the giant structure |
| Very high melting point (>3500 °C) | An enormous number of strong covalent bonds must be broken |
| Does not conduct electricity | All outer electrons are used in bonding; there are no free electrons |
| Insoluble in water | Giant non-polar covalent structure |
Uses: cutting tools and drill bits (hardest natural substance), abrasives, gemstones.
Graphite
Graphite is also a carbon allotrope, but with a very different structure. Each carbon atom forms three covalent bonds to three neighbours in flat hexagonal layers. The fourth outer electron per carbon is delocalised and free to move between layers.
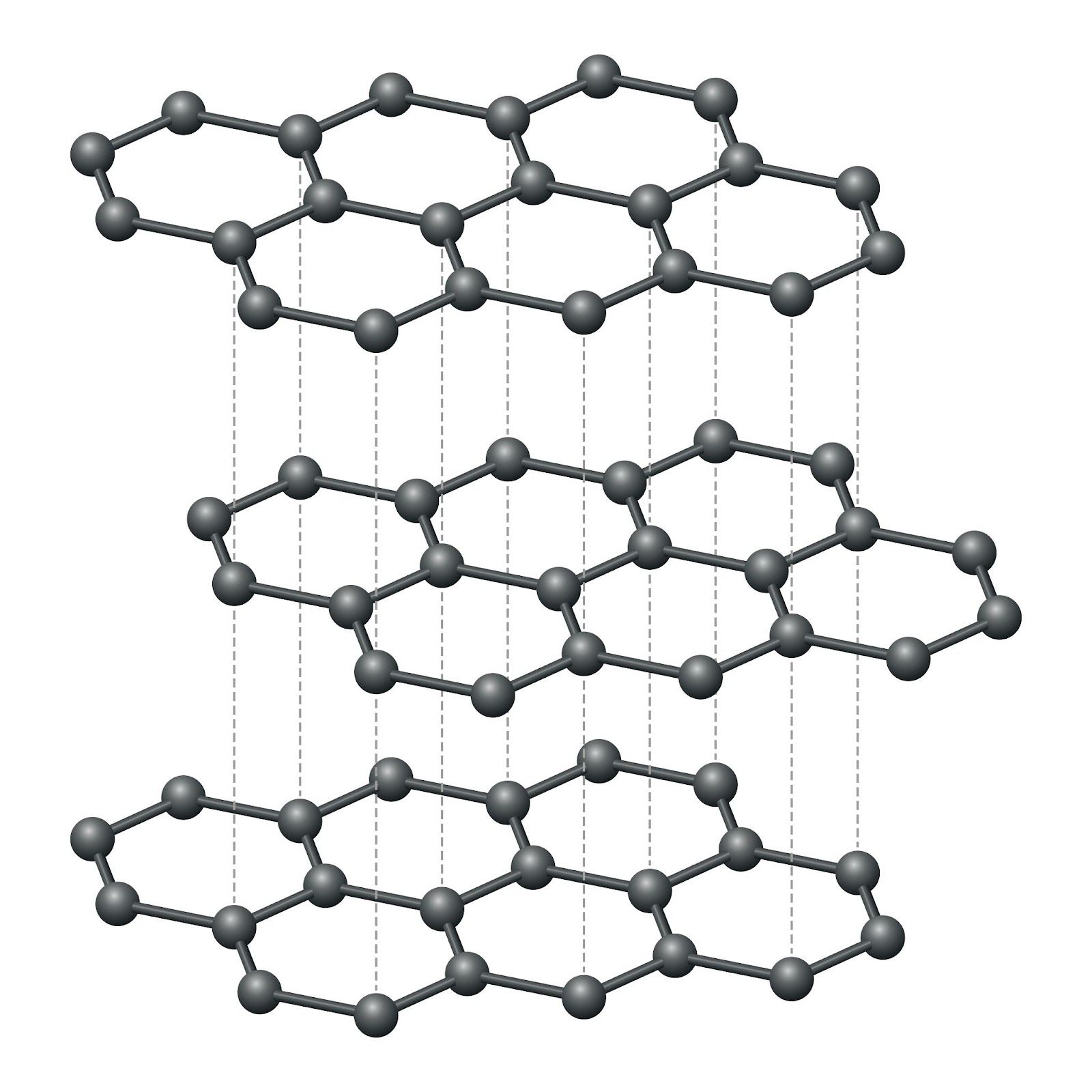
| Property | Reason |
|---|---|
| Soft and slippery | Layers are held together only by weak van der Waals forces and slide over each other easily |
| High melting point | Strong covalent bonds within each layer require considerable energy to break |
| Conducts electricity | Delocalised electrons flow through the structure and carry charge |
| Insoluble in water | Giant covalent structure |
Uses: lubricant (especially at high temperatures where oils break down), electrodes in electrolysis cells and batteries, pencil leads (mixed with clay).
Remember
The key contrast: diamond has every carbon bonded to four others — no free electrons, maximum hardness, no conductivity. Graphite has every carbon bonded to three others — one electron delocalised per carbon, soft slippery layers, conducts electricity.
Allotropy
Allotropy is the existence of an element in two or more structurally different forms in the same physical state.
Diamond and graphite are both pure carbon in the solid state — both consist entirely of carbon atoms with only covalent bonds. The difference is entirely in how those atoms are arranged and connected. Because the arrangement differs, the properties are dramatically different despite being the same element.