Atomic Structure
Matthew Williams
||9 min readAtomic StructureCSEC ChemistryElectronic ConfigurationIsotopesPaper 01Paper 02Section A
Subatomic particles and their properties, atomic number, mass number, atomic notation, electronic configuration for elements 1 to 20, ions, isotopes, relative atomic mass, and uses of radioactive isotopes.
All chemical behaviour — bonding, reactivity, conductivity, physical properties — traces back to atomic structure. Understanding what atoms are made of and how their electrons are arranged is the foundation for bonding, periodicity, and almost every other topic in chemistry.
The Atom
An atom is the smallest particle of an element that retains the chemical properties of that element. Every atom has two main regions:
- a dense central nucleus containing protons and neutrons
- a surrounding region of shells (energy levels) where electrons are found
Subatomic Particles
| Particle | Relative charge | Relative mass | Location |
|---|---|---|---|
| Proton | +1 | 1 | Nucleus |
| Neutron | 0 | 1 | Nucleus |
| Electron | −1 | 1/1840 | Shells around nucleus |
The mass of an electron is negligible compared to a proton or neutron. Nearly all of an atom's mass is concentrated in the nucleus. In a neutral atom, the number of protons equals the number of electrons, so the positive and negative charges cancel exactly.
Atomic Number and Mass Number
The atomic number (Z) is the number of protons in an atom. It uniquely identifies the element — every chlorine atom has exactly 17 protons, and no other element does. In a neutral atom, the atomic number also equals the number of electrons.
The mass number (A) is the total count of protons and neutrons in the nucleus:
The number of neutrons is therefore:
Example
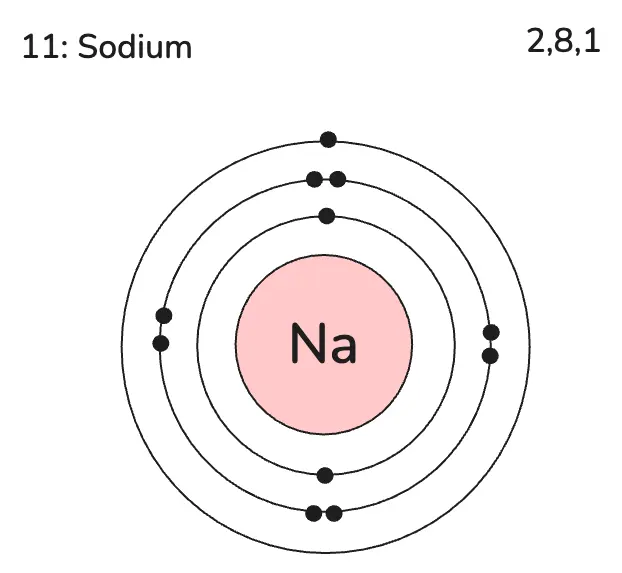
Sodium has atomic number 11 and mass number 23.
- Protons: 11
- Electrons: 11 (neutral atom)
- Neutrons: 23 − 11 = 12
Atomic Notation
The standard nuclear notation places the mass number at the top left and the atomic number at the bottom left of the element symbol:
So sodium is and chlorine-35 is .
When the species is an ion, the ionic charge appears at the top right. For example, with atomic number 12 has lost two electrons, leaving it with 12 protons but only 10 electrons. The ion has gained two electrons, so it has 8 protons and 10 electrons.
Exam Tip
When a question shows an ion, always start from the atomic number for proton count, then adjust the electron count by the charge. A 3+ ion has three fewer electrons than the neutral atom; a 2− ion has two more.
Electronic Configuration
Electrons occupy shells at increasing distances from the nucleus. Lower shells fill first. For elements up to atomic number 20, the shell capacities are:
| Shell | Maximum electrons |
|---|---|
| 1st (innermost) | 2 |
| 2nd | 8 |
| 3rd | 8 |
| 4th | 2 |
Electronic configuration is written as the number of electrons in each shell from innermost outward, separated by commas.
| Element | Symbol | Z | Configuration |
|---|---|---|---|
| Hydrogen | H | 1 | 1 |
| Helium | He | 2 | 2 |
| Lithium | Li | 3 | 2,1 |
| Beryllium | Be | 4 | 2,2 |
| Boron | B | 5 | 2,3 |
| Carbon | C | 6 | 2,4 |
| Nitrogen | N | 7 | 2,5 |
| Oxygen | O | 8 | 2,6 |
| Fluorine | F | 9 | 2,7 |
| Neon | Ne | 10 | 2,8 |
| Sodium | Na | 11 | 2,8,1 |
| Magnesium | Mg | 12 | 2,8,2 |
| Aluminium | Al | 13 | 2,8,3 |
| Silicon | Si | 14 | 2,8,4 |
| Phosphorus | P | 15 | 2,8,5 |
| Sulfur | S | 16 | 2,8,6 |
| Chlorine | Cl | 17 | 2,8,7 |
| Argon | Ar | 18 | 2,8,8 |
| Potassium | K | 19 | 2,8,8,1 |
| Calcium | Ca | 20 | 2,8,8,2 |
The electrons in the outermost shell are called valence electrons. They govern bonding and reactivity. The number of valence electrons corresponds to the group number in the periodic table; the number of occupied shells corresponds to the period.
Remember
Most atoms achieve stability by achieving a full outer shell — 8 electrons for most elements (the octet rule). Hydrogen and helium are stable with just 2 electrons (the duet rule). This drive for stability is why atoms form bonds.
Ions
An ion is a charged particle formed when a neutral atom gains or loses electrons. The proton count never changes — only the electron count changes.
Cations (positive ions) form when atoms lose electrons. Metals typically do this because their valence electrons are loosely held.
Anions (negative ions) form when atoms gain electrons. Non-metals typically do this because their outer shell is nearly full and the nucleus can attract additional electrons.
| Ion | Neutral atom configuration | Change | Ion configuration |
|---|---|---|---|
| Na⁺ | 2,8,1 | loses 1 electron | 2,8 |
| Mg²⁺ | 2,8,2 | loses 2 electrons | 2,8 |
| Al³⁺ | 2,8,3 | loses 3 electrons | 2,8 |
| Cl⁻ | 2,8,7 | gains 1 electron | 2,8,8 |
| O²⁻ | 2,6 | gains 2 electrons | 2,8 |
| N³⁻ | 2,5 | gains 3 electrons | 2,8 |
Each ion achieves the electron arrangement of the nearest noble gas.
Isotopes
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. They have the same atomic number but different mass numbers.
Because isotopes have the same number of electrons in the same arrangement, they have identical chemical properties. Physical properties such as density and diffusion rate differ because the atoms have different masses.
Example
The three naturally occurring isotopes of carbon:
| Isotope | Protons | Neutrons | Mass number |
|---|---|---|---|
| Carbon-12 () | 6 | 6 | 12 |
| Carbon-13 () | 6 | 7 | 13 |
| Carbon-14 () | 6 | 8 | 14 |
All three form exactly the same compounds and react identically.
Exam Tip
Isotopes differ in neutron number. Ions differ in electron number. These are the two most commonly confused definitions on Paper 01 questions about atomic structure.
Relative Atomic Mass
The relative atomic mass () of an element is the weighted average mass of its naturally occurring isotopes, expressed relative to one-twelfth the mass of a carbon-12 atom.
Carbon-12 is the international standard — its mass is defined as exactly 12, and all other atomic masses are measured against it.
When an element is a mixture of isotopes, its is not a whole number. Chlorine is approximately 75% Cl-35 and 25% Cl-37; the weighted average is about 35.5. This is why the periodic table shows 35.5 for chlorine rather than 35 or 37.
Uses of Radioactive Isotopes
Some isotopes are radioactive — their nuclei are unstable and emit radiation as they decay. This property makes them useful in several practical applications:
| Use | Example isotope | Principle |
|---|---|---|
| Carbon dating | Carbon-14 | Living organisms continuously absorb C-14; after death it decays at a known rate, allowing the age of organic material to be calculated |
| Cancer treatment (radiotherapy) | Cobalt-60 | High-energy radiation is directed at a tumour to destroy cancer cells |
| Medical tracers | Technetium-99m | A radioactive substance is introduced into the body and followed using a scanner to locate disease or abnormalities |
| Nuclear energy generation | Uranium-235 | Controlled fission releases large amounts of heat, which is used to generate electricity |
| Pacemakers (older designs) | Plutonium-238 | Provides a long-lived internal power source without needing external batteries |
Exam Tip
The syllabus requires at least three uses. Carbon dating, radiotherapy, and tracers are the three most reliably tested. Know the principle behind each, not just the name of the isotope.